Basic's of Chemistry
Learning objectives
- Recall how atoms, molecules and ions are bonded (energy holding them together)
- Classifying pure substances, elements on the Periodic Table
- Categorise the physical and chemical properties of various kitchen and cosmetic substances
Atoms, molecules and ions
ATOMS
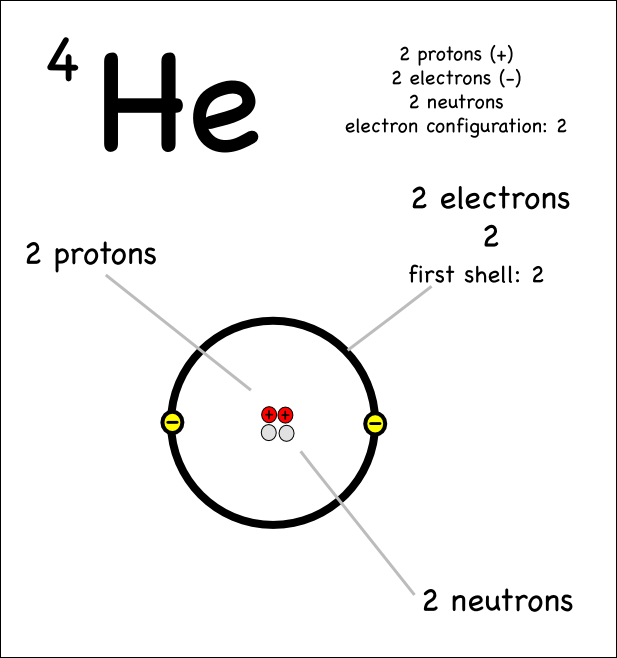
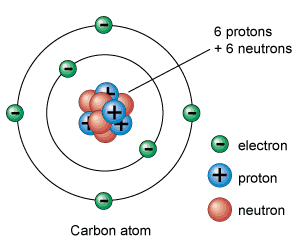
Atoms are the basic units of matter and the defining structure of elements. Atoms are made up of three particles: protons, neutrons and electrons.
energy charge of atoms:
protons = positive charge
neutrons= neutral (or no) charge
electrons= negative charge
refer to the carbon atom diagram >>
MOLECULES
If you want to build molecules, you will need atoms of different elements. Elements are the alphabet in the language of molecules. Each element is a little bit different from the rest.
ELEMENTS
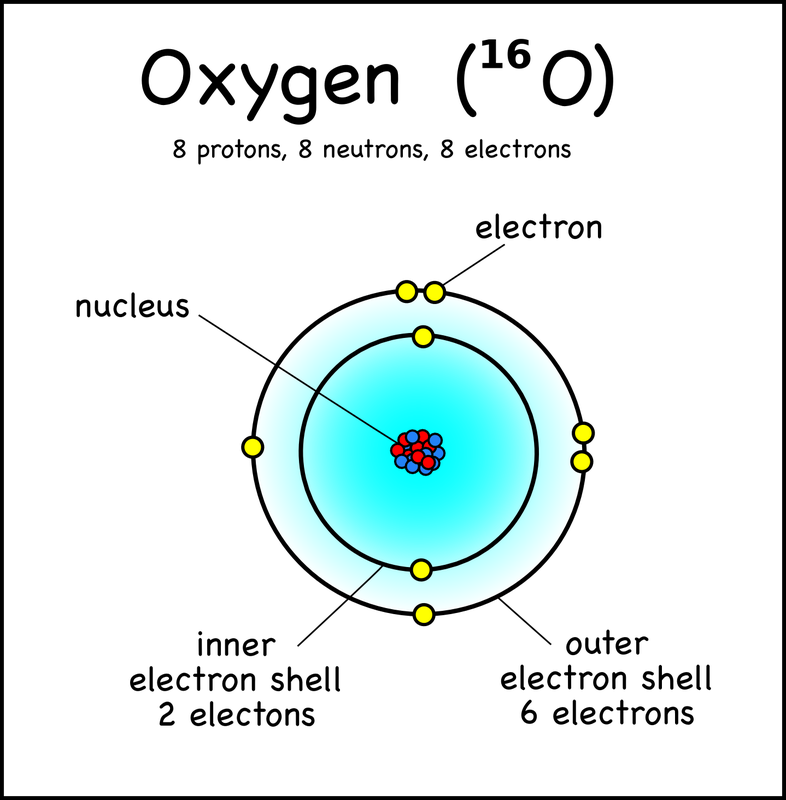
All of the atoms are made of the same basic sub-particles (protons, electrons and neutrons), but they are organised in different ways to make unique elements.
refer to the below images to see how each element is different just by changing what makes an atom.
WHAT IS AN ION?
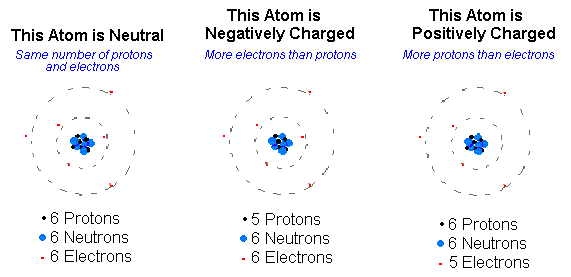
elements are stable because they have the SAME number of electrons and protons so overall they are NEUTRAL in charge.
Things happen to atoms where they may loose or gain electrons
Ions are electrically charged particles formed when atoms lose or gain electrons
when an atom gains an electron it will have more electrons than protons. This would mean it has an overall NEGATIVE charge
when an atoms lose an electron it will have more protons than electrons. This would mean it has an overall POSITIVE charge
Things happen to atoms where they may loose or gain electrons
Ions are electrically charged particles formed when atoms lose or gain electrons
when an atom gains an electron it will have more electrons than protons. This would mean it has an overall NEGATIVE charge
when an atoms lose an electron it will have more protons than electrons. This would mean it has an overall POSITIVE charge
So what do we call these positive and negatively charged atoms?
Overall positive = Cation
Overall negative = Anion
A good way to memorise this is to think of how cats have paws. Relate it to how "Cat"ions are "Pos"itive. See what I mean? Then all you have left is negative and anions if you can remember the first one
or anion - A -Negative -Ion
Overall positive = Cation
Overall negative = Anion
A good way to memorise this is to think of how cats have paws. Relate it to how "Cat"ions are "Pos"itive. See what I mean? Then all you have left is negative and anions if you can remember the first one
or anion - A -Negative -Ion